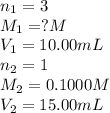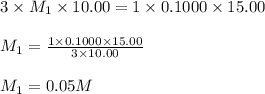Chemistry, 03.06.2021 04:30 elissiashontelbrown
g 10.00 mL of phosphoric acid (H3PO4) are titrated with 0.1000 M sodium hydroxide. 15.00 mL of the sodium hydroxide solution are used in this experiment. Determine the molarity of phosphoric acid.

Answers: 2


Other questions on the subject: Chemistry




Chemistry, 23.06.2019 09:30, Adolfosbaby
If the solubility of a gas in water is 1.22g/2.75 atm, what is it’s solubility (in g/l) at 1.0 atm
Answers: 1
You know the right answer?
g 10.00 mL of phosphoric acid (H3PO4) are titrated with 0.1000 M sodium hydroxide. 15.00 mL of the s...
Questions in other subjects:


Biology, 23.01.2020 21:31


Mathematics, 23.01.2020 21:31





Mathematics, 23.01.2020 21:31

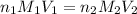 ........(1)
........(1)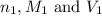 are the n-factor, molarity and volume of acid that is
are the n-factor, molarity and volume of acid that is 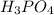
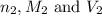 are the n-factor, molarity and volume of the base that is NaOH
are the n-factor, molarity and volume of the base that is NaOH