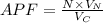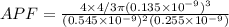Consider a hypothetical metal that has a density of 3.55 g/cm3, an atomic weight of 40.48 g/mol, and an atomic radius of 0.135 nm. Compute the atomic packing factor if the unit cell has tetragonal symmetry, values for the a and c lattice parameters are 0.545 and 0.255, respectively.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, rigobertogarza2
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 14:30, davidrodriguez122001
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 22.06.2019 16:50, Pookiev
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1
You know the right answer?
Consider a hypothetical metal that has a density of 3.55 g/cm3, an atomic weight of 40.48 g/mol, and...
Questions in other subjects:

Mathematics, 18.12.2020 08:20

Mathematics, 18.12.2020 08:20




Biology, 18.12.2020 08:20

Mathematics, 18.12.2020 08:20

Mathematics, 18.12.2020 08:20

Mathematics, 18.12.2020 08:20