
Chemistry, 01.06.2021 16:00 dededese2403
112 g of aluminum carbide react with 174 g water to produce methane and aluminum hydroxide in the reaction shown below.
2 Al4C3(s) + 12 H2O(l) → 3 CH4(g) + 4 Al(OH)3(s)
If aluminum carbide is the limiting reactant, how many moles of the excess reactant are left over
a
37.3 mol
b
4.68 mol
c
7.33 mol
d
131.94 mol

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 01:30, Nathaliasmiles
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2

Chemistry, 23.06.2019 06:40, 1991987
1.) which of the following is a molecule but not a compound? a. he b. f2 c. h2o d. ch4 2.) what is a physical combination of substances? a. a compound b. a molecule c. a mixture d. an element 3.) what is a chemical combination of substances? a. a compound b. an atom c. a mixture d. an element 4.) what is the relationship between the solute and solvent in a solution? a. they form a compound b. they form a mixture c. they form molecules d. they form chemical bonds 5.) the gases in air dissolve in water. what would be one way to reduce the amount of a gas dissolved in water? a. add more water b. reduce the air pressure c. increase the air pressure d. stir the water 6.) how would you determine the solubility of a substance? a. find how well it dissolved various substances. b. find the mass and the volume of the substance. c. find the temperature at which the substance evaporated. d. find how much i was able to dissolve in a solute. 7.) the periodic table organizes all of the kinds of a. molecules. b. compounds. c. atoms. d. ions. 8.)what distinguishes two substances combined to become a compound vs. two substances combined to become a mixture? a. whether they can be easily separated b. whether they chemically bond together c. whether they both are visible d. whether they are heterogeneous 9.) the principle components of air are: n2 78% o2 21% ar 0.95% co2 0.038% this is a solution of a. molecules and atoms. b. molecules. c. compounds and molecules. d. atoms.
Answers: 1
You know the right answer?
112 g of aluminum carbide react with 174 g water to produce methane and aluminum hydroxide in the re...
Questions in other subjects:

Mathematics, 11.01.2021 21:40

Biology, 11.01.2021 21:40


Mathematics, 11.01.2021 21:40

Mathematics, 11.01.2021 21:40

Mathematics, 11.01.2021 21:40


English, 11.01.2021 21:40


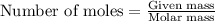 .....(1)
.....(1)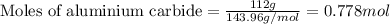
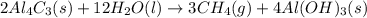
 of water
of water


