Chemistry, 31.05.2021 15:20 purplefive85
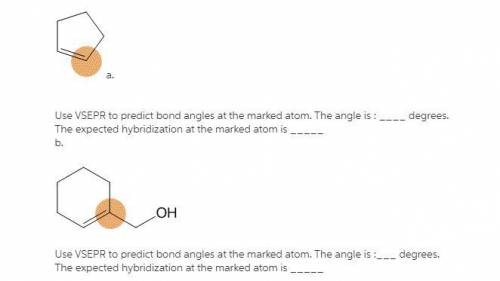
a. Use VSEPR to predict bond angles at the marked atom. The angle is : degrees. The expected hybridization at the marked atom is . b. Use VSEPR to predict bond angles at the marked atom. The angle is : degrees. The expected hybridization at the marked atom is .

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:30, Sbeech7246
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2

Chemistry, 22.06.2019 17:00, calmicaela12s
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1
You know the right answer?
a. Use VSEPR to predict bond angles at the marked atom. The angle is : degrees. The expected hybrid...
Questions in other subjects:

Mathematics, 09.12.2020 01:20




Mathematics, 09.12.2020 01:20



Physics, 09.12.2020 01:20

Social Studies, 09.12.2020 01:20