
Chemistry, 31.05.2021 06:00 Mintfu2580
How many moles of Fe(OH), are produced when 85.0 L of iron(III)
sulfate at a concentration of 0.600 mol/L reacts with excess
NaOH?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, alexisgoss8091
Which traits do human embryos have that link them to a common ancestor with fish and reptiles? a. scales and tail b. gill slits and scales c. tail and gill slits d. hair and tail
Answers: 2

Chemistry, 21.06.2019 23:20, anggar20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 14:50, wcraig1998
Complete the following statements to describe solids, liquids, and gases. select the correct answer from each drop-down menu. a solid a definite volume and a definite shape. a liquid a definite volume and a definite shape. a gas a definite volume and a definite shape
Answers: 1

Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
How many moles of Fe(OH), are produced when 85.0 L of iron(III)
sulfate at a concentration of 0.600...
Questions in other subjects:





Biology, 20.11.2020 17:00

Physics, 20.11.2020 17:00


Mathematics, 20.11.2020 17:00

Physics, 20.11.2020 17:00

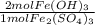 = 102 mol Fe(OH)₃
= 102 mol Fe(OH)₃


