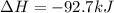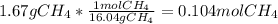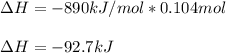Chemistry, 29.05.2021 17:00 allenpaietonp9v8sv
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If a 1.67 g sample of methane is burned at constant pressure, what will be the value of ∆H

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, logan12345677885675
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2


Chemistry, 23.06.2019 02:40, sherlock19
How can a mixture of salt water be separated into salt and water
Answers: 1

Chemistry, 23.06.2019 08:00, Robinlynn228
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 1
You know the right answer?
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If...
Questions in other subjects:

History, 09.10.2019 05:00

Mathematics, 09.10.2019 05:00


Biology, 09.10.2019 05:00


Biology, 09.10.2019 05:00


History, 09.10.2019 05:00