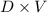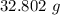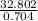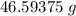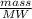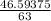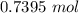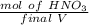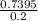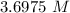Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, babygirl2984
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 21.06.2019 22:00, penelopymorales24
Match each object to its description: a. coma of a comet b. comet tail c. oort cloud haze surrounding a nucleus created by solar wind. hypothetical sphere around the solar system
Answers: 1

Chemistry, 22.06.2019 12:10, kaitlynbernatz2778
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3
You know the right answer?
A solution is made by adding 23.1 mL of concentrated nitric acid ( 70.4 wt% , density 1.42 g/mL ) to...
Questions in other subjects:

Mathematics, 01.02.2021 07:50

History, 01.02.2021 07:50


Biology, 01.02.2021 07:50

Mathematics, 01.02.2021 07:50




Mathematics, 01.02.2021 07:50

Social Studies, 01.02.2021 07:50