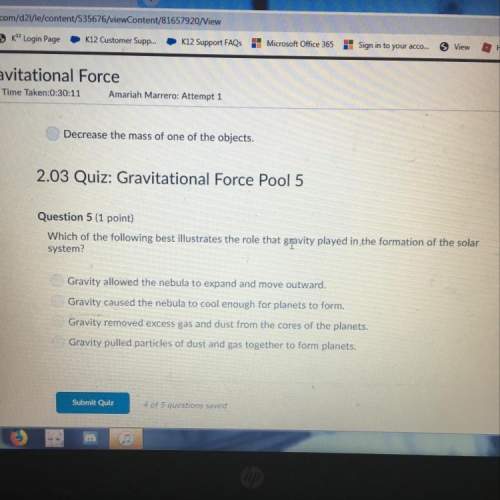
Chemistry, 27.05.2021 01:30 natd299736
A student made a sketch of a potential energy diagram to represent a reaction with a −ΔH value. A curve line graph is shown. The y axis of the graph has the title Potential Energy and kJ written in parenthesis. The x axis of the graph has the title Reaction Pathway. The curve begins at a higher level and ends at a slightly lower level. A broken horizontal line is shown from a point labelled X on the y axis to the point where the curve begins. Another broken horizontal line is shown from a point labeled Y on the y axis to the point where the curve ends. Explain, using complete sentences, why the diagram made by the student is correct or incorrect. Be sure to also explain what the values of X and Y represent.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, aubreykenzie686
Naoki's bicycle has a mass of 10 kg. if naoki sits on her bicycle and starts pedaling with a force of 168 n, causing an acceleration of 2.8 m/s2, what is naoki's mass?
Answers: 1


Chemistry, 22.06.2019 22:30, itsmaddierae11
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1
You know the right answer?
A student made a sketch of a potential energy diagram to represent a reaction with a −ΔH value. A cu...
Questions in other subjects:



Social Studies, 17.12.2020 22:20

Biology, 17.12.2020 22:20


Mathematics, 17.12.2020 22:20

Biology, 17.12.2020 22:20