
Chemistry, 26.05.2021 18:30 unknown6669
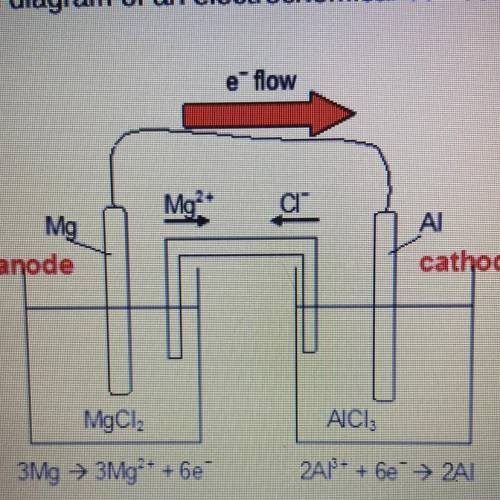
Which observation would be most likely to happen if a power supply were added to the electrochemical cell?
A)Magnesium would be oxidized.
B)Electrons would flow through the salt bridge.
C)The Al electrode would become neutral.
D)The Mg electrode would become the cathode.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, jbarbie3
12. complete each of the following word equations for synthesis reactions. a. sodium + oxygen -> b. magnesium + fluorine -> 13. complete and balance the equations for the decomposition reactions. a. hgo -> [with the triangle heat symbol above the arrow] b. h2o(l) -> [with "electricity" written above the arrow]
Answers: 1


Chemistry, 22.06.2019 14:50, rebeccamckellpidge
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 21:00, agarcia24101993
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
Which observation would be most likely to happen if a power supply were added to the electrochemical...
Questions in other subjects:



Biology, 06.10.2019 17:30

Biology, 06.10.2019 17:30


Mathematics, 06.10.2019 17:30

Biology, 06.10.2019 17:30


History, 06.10.2019 17:30



