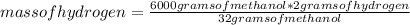Chemistry, 24.05.2021 07:30 bobiscool3698
Determine what mass of carbon monoxide and what mass of hydrogen are required to form 6.0 kg of methanol by the reaction CO(g) + 2H2(g) -> C H3OH(l)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:50, trinityrae4657
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 20:00, emilyswinge4421
Listenbase your answer to the question on the information below. nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body. cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment. which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2


Chemistry, 23.06.2019 09:00, blossie94681
The vapor pressure of water at 25.0°c is 23.8 torr. determine the mass of glucose (molar mass = 180 g/mol) needed to add to 500.0 g of water to change the vapor pressure to 22.8 torr.
Answers: 1
You know the right answer?
Determine what mass of carbon monoxide and what mass of hydrogen are required to form 6.0 kg of meth...
Questions in other subjects:

English, 26.01.2021 19:40


History, 26.01.2021 19:40


Spanish, 26.01.2021 19:40

History, 26.01.2021 19:40


Spanish, 26.01.2021 19:40

History, 26.01.2021 19:40