Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, homeschool0123
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 09:00, 2024cynthiatercero
Which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 3

Chemistry, 22.06.2019 11:30, ansuaprajita1506
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 12:00, luffybunny
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1
You know the right answer?
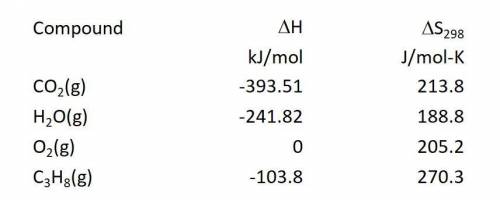
Calculate the entropy change in J/mol-K for the reaction: C3H8(g) + 5O2(g) ---> 3CO2(g) + 4H2O(g)...
Questions in other subjects:

Biology, 17.11.2019 20:31

Mathematics, 17.11.2019 20:31

Mathematics, 17.11.2019 20:31




Social Studies, 17.11.2019 20:31

Mathematics, 17.11.2019 20:31


History, 17.11.2019 20:31