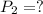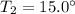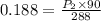Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1


Chemistry, 23.06.2019 06:00, hopechinn6646
Complete the sentences to best explain the ranking. match the words below to the appropriate blanks in the sentences. a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1

Chemistry, 23.06.2019 07:00, jennnifercrd59jc
Choose the correct statement about licensed veterinarians in the united states. a. they must be certified by the avma. b. they can treat all nonhuman animals. c. they can can treat only animals specified on the license. d. they must choose a specialty.
Answers: 2
You know the right answer?
Please help
The pressure of a 70.0L sample of gas is 600 mm Hg at 20.0°C. If the temperature drops...
Questions in other subjects:

English, 29.08.2019 09:00

History, 29.08.2019 09:00



Mathematics, 29.08.2019 09:00




History, 29.08.2019 09:00