
Chemistry, 20.05.2021 07:10 lindseylewis313
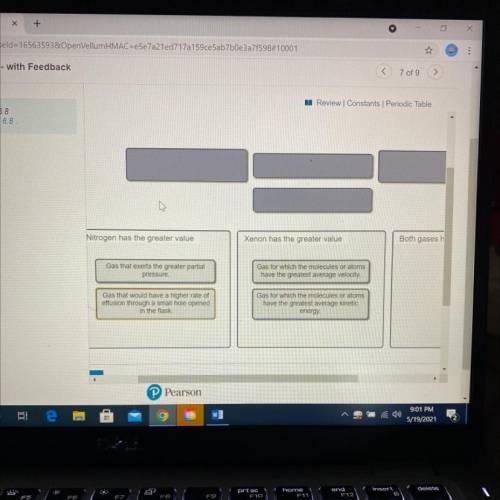
A flask at room temperature contains exactly equal amounts (in moles) of nitrogen and xenon.
Sort the conditions based on the gas described.
Drag the appropriate items to their respective bins.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, deaishaajennings123
What is the equilibrium constant of aa+bb=cc+dd
Answers: 1


Chemistry, 23.06.2019 00:30, motorxr714
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
A flask at room temperature contains exactly equal amounts (in moles) of nitrogen and xenon.
Sort t...
Questions in other subjects:


Mathematics, 31.01.2020 15:56


Mathematics, 31.01.2020 15:56


Biology, 31.01.2020 15:56

English, 31.01.2020 15:56


World Languages, 31.01.2020 15:56

Social Studies, 31.01.2020 15:56



