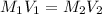Chemistry, 20.05.2021 01:00 MoneyMike42
A 72.4 mL solution of Cu(OH) is neutralized by 47.8 mL of a 0.56 M H2(C204) solution. What is the concentration of the Cu(OH)?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:10, apowers6361
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 13:00, carlinryan
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 15:20, mydoggy152
Fossil fuels are organic compounds that are made from
Answers: 1

Chemistry, 23.06.2019 07:00, asims13
The following transition occurs at a molecular level for a substance. what transition corresponds to this change in microscopic structure? the carbon dioxide molecules on the left are in a regular, tightly packed pattern. after heating, it becomes much lower density. a. melting b. boiling c. sublimation d. freezing
Answers: 1
You know the right answer?
A 72.4 mL solution of Cu(OH) is neutralized by 47.8 mL of a 0.56 M H2(C204) solution. What is the co...
Questions in other subjects:


Mathematics, 24.09.2020 14:01



Mathematics, 24.09.2020 14:01



Engineering, 24.09.2020 14:01

Biology, 24.09.2020 14:01

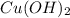 is 0.369 M.
is 0.369 M. = 72.4 mL,
= 72.4 mL,  = ?
= ?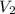 = 47.8 mL,
= 47.8 mL,  = 0.56 M
= 0.56 M