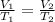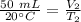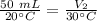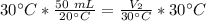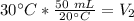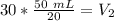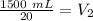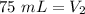Chemistry, 18.05.2021 04:50 meredith48034
1.
At constant pressure. 50 milliliters (mL) of a gas
at 20°C is heated to 30° C. The new volume of
the gas in milliliters (ml) is equal

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, lazavionadams81
Identify which properties could correspond to solids, plasmas, or both. maintain a unique shape. collide infrequently with other particles. have very high velocities. conduct electricity. protons. have a low temperature. has long-range order.
Answers: 1

Chemistry, 22.06.2019 04:00, nikkih1225
Electric charge is what ? a. kinetic energy b. radiation c. discovery d. electricity
Answers: 1

Chemistry, 22.06.2019 13:30, annanikherrera
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 16:30, montanolumpuy
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3
You know the right answer?
1.
At constant pressure. 50 milliliters (mL) of a gas
at 20°C is heated to 30° C. The new vol...
at 20°C is heated to 30° C. The new vol...
Questions in other subjects:










Advanced Placement (AP), 29.07.2019 09:00