
Chemistry, 14.05.2021 23:30 coxtinam16
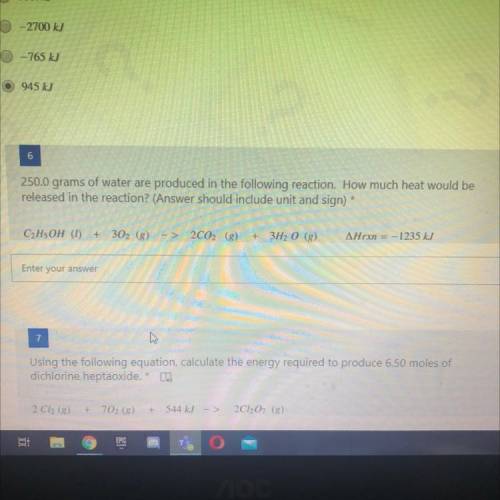
250.0 grams of water are produced in the following reaction. How much heat would be
released in the reaction? (Answer should include unit and sign)
+
>
C2H5OH (1)
Arxn = -1235 kJ
+
302 (8)
2CO2 (g)
3H2O ()

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:00, mercymain1014
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1
You know the right answer?
250.0 grams of water are produced in the following reaction. How much heat would be
released in the...
Questions in other subjects:


Physics, 31.08.2019 03:00







Mathematics, 31.08.2019 03:00

Mathematics, 31.08.2019 03:00



