
Chemistry, 14.05.2021 20:10 jazzycintron14
Geologists can estimate the age of rocks by their uranium-238 content. The uranium is incorporated in the rock as it hardens and then decays with first-order kinetics and a half-life of 4.5 billion years. A rock is found to contain 83.9% of the amount of uranium-238 that it contained when it was formed. (The amount that the rock contained when it was formed can be deduced from the presence of the decay products of U-238.) How old is the Rock?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:50, deanlmartin
Choose all that apply. when creating a graph, you should: determine the x- and y- variables label the scale on the x- and y- axes plot the data points draw a line of best fit to represent the data trend
Answers: 1


Chemistry, 22.06.2019 06:30, yolo123321
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2
You know the right answer?
Geologists can estimate the age of rocks by their uranium-238 content. The uranium is incorporated i...
Questions in other subjects:


Mathematics, 10.02.2022 15:50

Mathematics, 10.02.2022 15:50


English, 10.02.2022 15:50


Mathematics, 10.02.2022 15:50

Mathematics, 10.02.2022 15:50


English, 10.02.2022 15:50

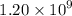 years
years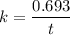
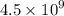 years
years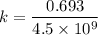
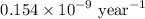
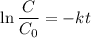
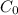 = initial amount
= initial amount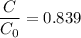
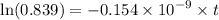
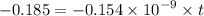
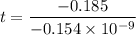
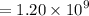 years
years

