2.How much excess reactant will be left?

Chemistry, 06.05.2021 21:20 roseemariehunter12
NEED HELP ASAP
1.Which is the limiting reactant?
2.How much excess reactant will be left?
3. How many moles of product can be made?
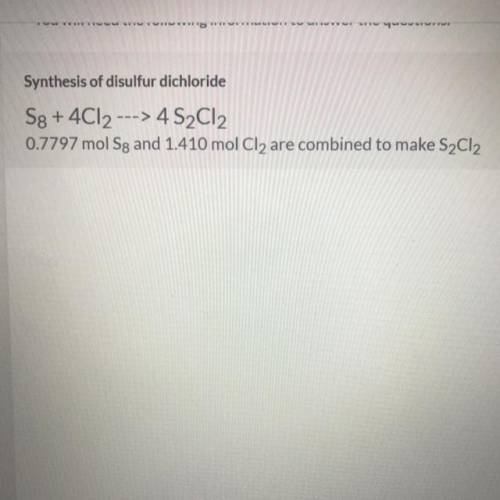

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, sandersmakaylaovq5vu
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 03:00, parisaidan366
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 06:30, dimondqueen511
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 10:50, lejeanjamespete1
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1
You know the right answer?
NEED HELP ASAP
1.Which is the limiting reactant?
2.How much excess reactant will be left?
2.How much excess reactant will be left?
Questions in other subjects:


History, 03.02.2021 01:50

Chemistry, 03.02.2021 01:50

Health, 03.02.2021 01:50

Mathematics, 03.02.2021 01:50


Mathematics, 03.02.2021 01:50

History, 03.02.2021 01:50




