
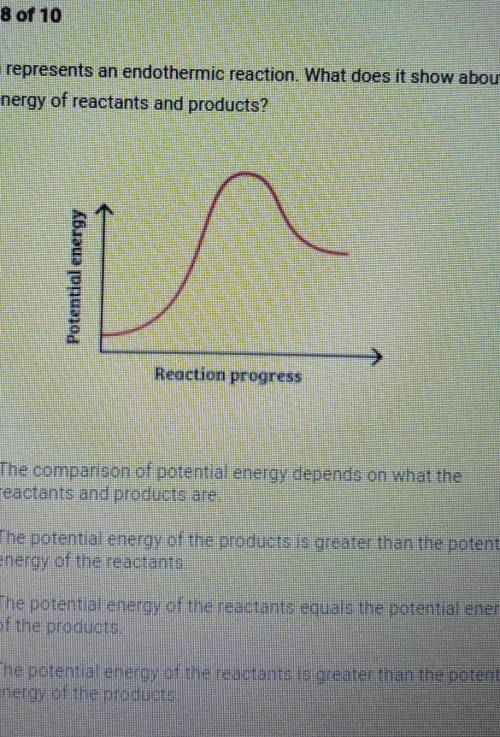
This graph represents an endothermic reaction. What does it show about the potential energy of reactants and products? Abuzua nunuad in Reaction progress A. The comparison of potential energy depends on what the reactants and products are, B. The potential energy of the products is greater than the potential energy of the reactants C. The potential energy of the reactants equals the potential energy of the products D. The potential energy of the reactants is greater than the potential energy of the products,

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:10, kellinvagneur
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1

Chemistry, 22.06.2019 20:00, 20calzoy
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
This graph represents an endothermic reaction. What does it show about the potential energy of react...
Questions in other subjects:

Mathematics, 14.05.2021 01:00

History, 14.05.2021 01:00



Mathematics, 14.05.2021 01:00


Mathematics, 14.05.2021 01:00

History, 14.05.2021 01:00





