
Chemistry, 02.05.2021 23:40 2023brewerantonio
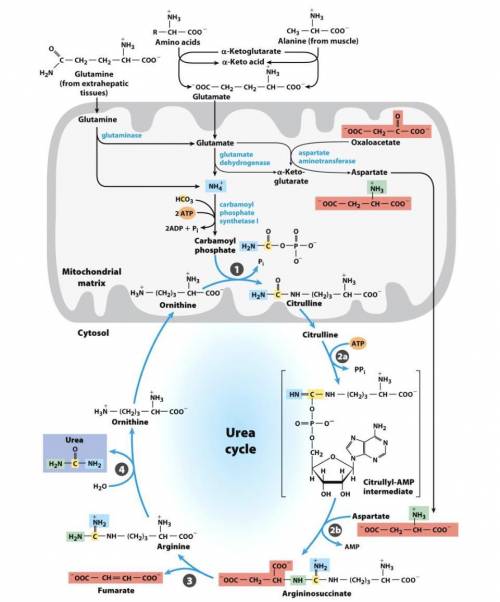
A. Write a series of balanced equations, and fill in the blanks for an overall equation, for the complete oxidation of 2 mol of glutamate to 2 mol of α-ketoglutarate and 1 mol of urea. Note that the overall equation (net reaction) does not include aspartate, fumarate, or oxaloacetate, so you must include balanced equations for the interconversion of these molecules. (Hint: what pathway converts fumarate to oxaloacetate?) For reference, the urea cycle figure is shown on the back. You will need a total of 9 to 11 reactions in order to generate the balanced overall equation.
Overall equation/net reaction (fill in stoichiometry):(The stoichiometry of water and H+is not important here, so don’t stress about that)2 glutamate + __CO2+ 4 H2O + __NAD++ __ATP →2 α-ketoglutarate + urea + ___NADH + 7 H++ __ADP + __AMP + __Pi+ __PPi
b. Based on the final equation above, does the process result in a net gain or loss of ATP equivalents? How many?

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:00, issachickadi
Which of the following is not a good technique for managing used oil? a) have specific, labeled catch pans available for technicians who are collecting oil b) spills in your shop and any releases on pavement or outside should be poured down a drain c) do not use oil containers for antifreeze or other non-similar fluids d) be prepared for oil spills with the proper absorbents
Answers: 1


Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3
You know the right answer?
A. Write a series of balanced equations, and fill in the blanks for an overall equation, for the com...
Questions in other subjects:


History, 30.07.2019 09:20

History, 30.07.2019 09:20

History, 30.07.2019 09:20

History, 30.07.2019 09:20


Biology, 30.07.2019 09:20


World Languages, 30.07.2019 09:20



