
Chemistry, 02.05.2021 01:00 lilzaya510
A model of tin, an element with the atomic number 50, is shown here. The valence electrons are modeled here in this image. Which statements are supported by the information in the model? Select ALL That apply.
A) Tin needs four more electrons to complete its outer shell.
B) Tin has no neutrons in the nucleus, as is shown in the model.
C) Tin is highly reactive because it only has four valence electrons.
D) Tin is negatively charged because it more electrons than protons.
E) Tin has a low reactivity because it has full inner shells of electrons.
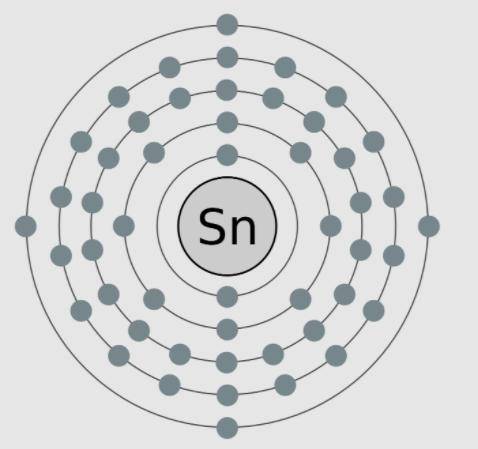

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:40, vpowell5371
Astudent made the lewis dot diagram of a compound shown. what is the error in the lewis dot diagram? a)an o atom should transfer all of its six electrons to mg because the formula is mgo b) both electrons of mg should be transferred to one o adam because the formula is mgo c) the electrons should be transferred from each o add him to capital mg because mg has fewer electrons d) the number of dots around mg should be four because it has to transfer two electrons to each o
Answers: 1


Chemistry, 22.06.2019 09:20, pandaman632
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1
You know the right answer?
A model of tin, an element with the atomic number 50, is shown here. The valence electrons are model...
Questions in other subjects:


Biology, 17.12.2019 14:31

Mathematics, 17.12.2019 14:31

Mathematics, 17.12.2019 14:31

Biology, 17.12.2019 14:31




Chemistry, 17.12.2019 14:31

Mathematics, 17.12.2019 14:31



