
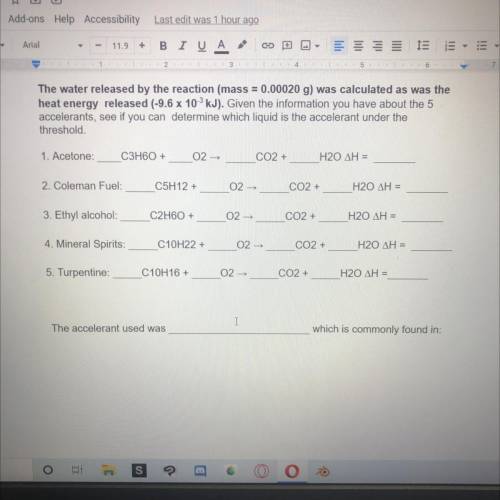
The water released by the reaction (mass = 0.00020 g) was calculated as was the
heat energy released (-9.6 x 10 kJ). Given the information you have about the 5
accelerants, see if you can determine which liquid is the accelerant under the
threshold.
1. Acetone:
C3H60+
02-
CO2 +
H20 ΔΗ =
2. Coleman Fuel:
C5H12 +
02-
CO2 +
H20 ΔΗ =
3. Ethyl alcohol
C2H60 +
02 -
CO2 +
H20 ΔΗ =
4. Mineral Spirits:
C10H22 +
02-
CO2 +
H20 AH =
5. Turpentine:
C10H16 +
02 -
CO2 +
H20 ΔΗ =
The accelerant used was
I
which is commonly found in:

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, madisonrosamond99
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 09:20, payshencec21
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 21:50, namoralessimon03
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1
You know the right answer?
The water released by the reaction (mass = 0.00020 g) was calculated as was the
heat energy releas...
Questions in other subjects:

History, 17.06.2021 16:00



Biology, 17.06.2021 16:00





Mathematics, 17.06.2021 16:00

Social Studies, 17.06.2021 16:00



