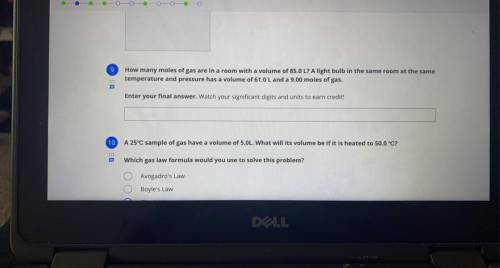
Question number 9 please!
...

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 14:00, claudia122752
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 18:30, lattimorekeonna1
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
You know the right answer?
Questions in other subjects:



Social Studies, 12.01.2021 23:50




Mathematics, 12.01.2021 23:50

English, 12.01.2021 23:50

Mathematics, 12.01.2021 23:50

Biology, 12.01.2021 23:50