
Chemistry, 22.04.2021 22:40 rikac72791
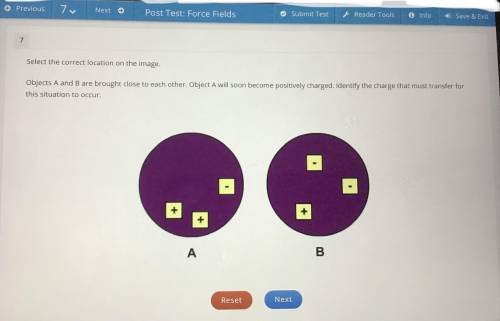
Objects A and B are brought close to each other. Object A will soon become positively charged. Identify the charge that must transfer for this situation to occur.
(You have to click one of the charges like the positive = + or the negative = -)
It’s definitely not the Object B positive charge. (I already clicked tht one this first time & got it wrong.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, brittanysanders
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 19:20, johnkings140
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

You know the right answer?
Objects A and B are brought close to each other. Object A will soon become positively charged. Ident...
Questions in other subjects:

English, 15.11.2019 22:31

History, 15.11.2019 22:31


Mathematics, 15.11.2019 22:31





English, 15.11.2019 22:31




