Chemistry, 20.09.2019 01:00 kotetravels10
Asample of cacl2⋅2h2o/k2c2o4⋅h2o solid salt mixture is dissolved in ~150 ml de-ionized h2o. the oven dried precipitate has a mass of 0.333 g. the limiting reactant in the salt mixture is k2c2o4⋅h2o. cacl2⋅2h2o(aq) + k2c2o4⋅h2o(aq) à cac2o4⋅h2o(s) + 2kcl(aq) + 2h2o(l) starting material (sm) product molar mass (mm) g/mol: cacl2⋅2h2o = 147.02 k2c2o4⋅h2o = 184.24 cac2o4 = 128.10 determine mass of k2c2o4⋅h2o(aq) in salt mixture in grams. answer to 3 places after the decimal and include unit, g

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, 10040813
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 22.06.2019 16:00, julesperez22
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 19:20, johnkings140
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1
You know the right answer?
Asample of cacl2⋅2h2o/k2c2o4⋅h2o solid salt mixture is dissolved in ~150 ml de-ionized h2o. the oven...
Questions in other subjects:


Mathematics, 08.12.2019 23:31


Biology, 08.12.2019 23:31




History, 08.12.2019 23:31


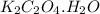 in the salt mixture is 0.424 grams.
in the salt mixture is 0.424 grams.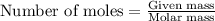 .....(1)
.....(1)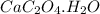 :
: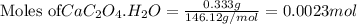
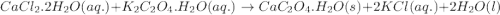
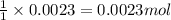 of
of