Consider the reaction below. c2h4(g) + h2(g) to c2h6(g) which change would likely cause the greatest increase in the rate of the reaction?
a decrease temperature and decrease pressure
b increase temperature and decrease pressure
c decrease temperature and increase pressure
d increase temperature and increase pressure

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, jocelynmarquillo1
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 05:00, mahhvelousg97
You mix the pks of succinic acid are 4.21 and 5.64. how many gramsa graduate student at sdsu wants to measure the activity of a particular enzyme at ph 4.0. to buffer her reaction, she will use a buffer system based on one of the acids listed below, which acid is most appropriate for the experiment? of monosodium succinate (fw = 140 g/mol) and disodium succinate (fw = 162 g/mol) must be added to 1 l of water to produce a solution with a ph 5.28 and a total solute concentration of 100 mm? (assume the total volume remains 1 liter, answer in grams monosodium succinate, grams disodium succinate, respectively.) volumes of 0.05 m nah2po4 and 0.05 m na2hpo4 (pk's for phosphoric acid are 2.15, 6.82 and 12.38). which of the following best describes the resulting solution?
Answers: 2


Chemistry, 23.06.2019 03:30, tamariarodrigiez
Which of the following describes the entropy change as a solution is made from a liquid and solid
Answers: 1
You know the right answer?
Consider the reaction below. c2h4(g) + h2(g) to c2h6(g) which change would likely cause the greatest...
Questions in other subjects:

English, 28.12.2019 06:31

Mathematics, 28.12.2019 06:31

English, 28.12.2019 06:31




Geography, 28.12.2019 06:31

English, 28.12.2019 06:31

Mathematics, 28.12.2019 06:31

Spanish, 28.12.2019 06:31

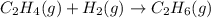 greatest increase in rate of reaction only occur when we increase the temperature and also increase the pressure.
greatest increase in rate of reaction only occur when we increase the temperature and also increase the pressure.