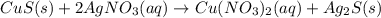Chemistry, 15.11.2019 20:31 Tringirl233
E. solid copper sulfide and silver nitrate react to form copper (ii) nitrate and solid silver sulfide. write a balanced chemical equation that describes the reaction. identify the oxidation number of each element in the reaction. (you do not need to include the total contribution of charge.) is this reaction a redox reaction or a non-redox reaction? explain your answer.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, Homepage10
Theoretically, which metal should be the most reactive? hydrogen lithium francium fluorine
Answers: 1

Chemistry, 22.06.2019 04:00, tifftifftiff5069
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 21:00, agarcia24101993
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
E. solid copper sulfide and silver nitrate react to form copper (ii) nitrate and solid silver sulfid...
Questions in other subjects:

Biology, 19.07.2019 00:20


Mathematics, 19.07.2019 00:20





Biology, 19.07.2019 00:20


Mathematics, 19.07.2019 00:20