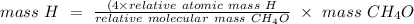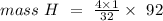Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 18:00, meowmeowcow
Find the mass, in grams, of 5.00*10^23 molecules of f2
Answers: 3


Chemistry, 23.06.2019 08:00, oopsorry
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1

Chemistry, 23.06.2019 10:00, jdmXdude3140
How to draw a diagram to represent a calcium metal lattice?
Answers: 3
You know the right answer?
How many grams of hydrogen are in 92 g of ch4o?...
Questions in other subjects:

Arts, 05.05.2020 07:50

Mathematics, 05.05.2020 07:50

English, 05.05.2020 07:50





History, 05.05.2020 07:50


Arts, 05.05.2020 07:50