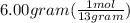Chemistry, 01.09.2019 10:00 MIYAISSAVGE2409
How many moles of atoms are in 6.00 g of 13c?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
How many moles of atoms are in 6.00 g of 13c?...
Questions in other subjects:




SAT, 31.03.2021 22:30

Computers and Technology, 31.03.2021 22:30


Mathematics, 31.03.2021 22:30


Mathematics, 31.03.2021 22:30