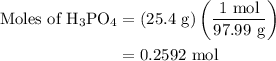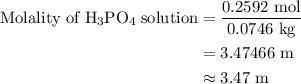Chemistry, 28.08.2019 16:00 smkw04p3ao0n
Calculate the molality of a 25.4% (by mass) aqueous solution of phosphoric acid (h3po4).

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, orlando19882000
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1

Chemistry, 21.06.2019 23:30, tylerineedhelp
Ihat will happen if i added baking soda to vinegar
Answers: 2

Chemistry, 22.06.2019 10:20, blondielocks2002
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 11:30, chelseychew32
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2
You know the right answer?
Calculate the molality of a 25.4% (by mass) aqueous solution of phosphoric acid (h3po4)....
Questions in other subjects:


Mathematics, 10.03.2022 09:00

Mathematics, 10.03.2022 09:00


Mathematics, 10.03.2022 09:00

Mathematics, 10.03.2022 09:00


English, 10.03.2022 09:00


Social Studies, 10.03.2022 09:00

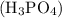 is
is 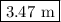 .
.
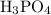 solution is as follows:
solution is as follows:
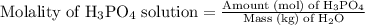 …… (1)
…… (1)
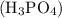 is 25.4 % by mass. So the mass of
is 25.4 % by mass. So the mass of 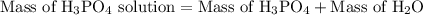 …… (2)
…… (2)
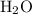 .
.
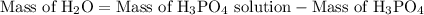 …… (3)
…… (3)
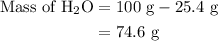
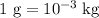
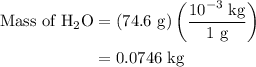
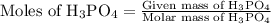 …… (4)
…… (4)