
Chemistry, 16.10.2019 12:00 weridness80
Determine the ph of a 0.05 m solution of hydrocyanic acid (hcn). hydrocyanic acid has an acid-dissociation constant of 4.9 × 10-10.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, fbillinton
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1



Chemistry, 22.06.2019 19:50, jakaylathomas11
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3
You know the right answer?
Determine the ph of a 0.05 m solution of hydrocyanic acid (hcn). hydrocyanic acid has an acid-dissoc...
Questions in other subjects:

Social Studies, 02.03.2021 04:30

Mathematics, 02.03.2021 04:30

Mathematics, 02.03.2021 04:30




History, 02.03.2021 04:30

Mathematics, 02.03.2021 04:30


Mathematics, 02.03.2021 04:30


![k_a=\frac{[H^+][CN^-]}{[HCN]}](/tpl/images/0324/9828/15aab.png)
![[H^+]](/tpl/images/0324/9828/07acb.png) and
and 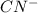 will be, 'x'
will be, 'x'


![pH=-\log [H^+]](/tpl/images/0324/9828/37e81.png)




