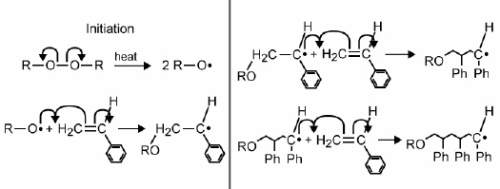
Consider the following diagram of a chemical reaction.
what is the product that is forme...

Chemistry, 26.01.2020 00:31 brianmcclain7010
Consider the following diagram of a chemical reaction.
what is the product that is formed in this reaction?
a polymer
a monomer
an alkyl halide
a carboxylic acid

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, scottbrandon653
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2


Chemistry, 22.06.2019 19:00, HaydenSturgis1
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 21:30, leenzazou587
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
Questions in other subjects:

Mathematics, 06.06.2020 20:58






Social Studies, 06.06.2020 20:58


Mathematics, 06.06.2020 20:58

Mathematics, 06.06.2020 20:58



