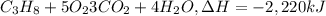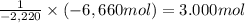Chemistry, 24.01.2020 19:31 nawafcanada
One mole of propane (44 grams) reacts with 5 moles of oxygen to produce 2220 kj of heat. how many grams of propane would produce 6660 kj of heat? a.220 g b. 132 g c. 4440 g d.49 g

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:50, daniel9299
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 10:40, justicejesusfreak
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

Chemistry, 22.06.2019 16:00, julesperez22
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3
You know the right answer?
One mole of propane (44 grams) reacts with 5 moles of oxygen to produce 2220 kj of heat. how many gr...
Questions in other subjects:





Mathematics, 30.06.2019 01:30

History, 30.06.2019 01:30




Mathematics, 30.06.2019 01:30