
Avogadro’s law and charles’s law describe a proportionality of the volume of a gas when the pressure is constant. which describes the proportionality that allows these laws to be combined when describing a gas?
volume is directly proportional to temperature (avogadro’s law) and to moles (charles’s law).
volume is inversely proportional to temperature (avogadro’s law) and to moles (charles’s law).
volume is directly proportional to moles (avogadro’s law) and to temperature (charles’s law).
volume is inversely proportional to moles (avogadro’s law) and to temperature (charles’s law).

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:00, emilyswinge4421
Listenbase your answer to the question on the information below. nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body. cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment. which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2



Chemistry, 24.06.2019 03:30, joshuanieto44
Why do we write chemical compounds inside brackets example: [ag (nh3)2]cl [zn (nh3)4](oh)2writing within and without does it matter? how, clear my doubt.
Answers: 1
You know the right answer?
Avogadro’s law and charles’s law describe a proportionality of the volume of a gas when the pressure...
Questions in other subjects:



Mathematics, 02.12.2020 08:40


Mathematics, 02.12.2020 08:40

Mathematics, 02.12.2020 08:40




Spanish, 02.12.2020 08:50

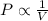 (At constant temperature and number of moles)
(At constant temperature and number of moles)
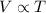 (At constant pressure and number of moles)
(At constant pressure and number of moles)
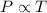 (At constant volume and number of moles)
(At constant volume and number of moles)
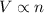 (At constant temperature and pressure)
(At constant temperature and pressure)

