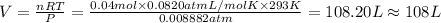Chemistry, 09.10.2019 21:30 ryantrajean7
What is the volume of 0.04 mol of a gas at 0.9 kpa and 20°c?
a. 108 l
b. 14.8 l
c. 87.5 l
d. 7.4 l

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 11:30, cjmckee2001
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3

Chemistry, 23.06.2019 11:50, natorihill629
Charles's law describes the relationship of the volume and temperature of gas at a constant mass and pressure. according to this law, what would happen to the temperature of the gas if its volume decreased from 1.0 l to 0.50 l?
Answers: 3

Chemistry, 23.06.2019 14:00, kcutler8603
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 14.0 mol cesium fluoride with 14.0 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1
You know the right answer?
What is the volume of 0.04 mol of a gas at 0.9 kpa and 20°c?
a. 108 l
b. 14.8 l
...
a. 108 l
b. 14.8 l
...
Questions in other subjects:

History, 02.12.2021 01:40


Mathematics, 02.12.2021 01:40

Spanish, 02.12.2021 01:40

Physics, 02.12.2021 01:40



Social Studies, 02.12.2021 01:40


Mathematics, 02.12.2021 01:40