
Chemistry, 08.01.2020 20:31 claudia122752
When sodium is excited in a flame, two ultraviolet spectral lines at lambda - 372.1 nm and lambda = 376.4 nm respectively are emitted . which wavelength represented photons?
a) higher energy?
b) longer wavelengths?
c) higher frequences?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, medlinalex
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 20:20, catchonyet
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1


Chemistry, 23.06.2019 04:00, ayoismeisjjjjuan
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2
You know the right answer?
When sodium is excited in a flame, two ultraviolet spectral lines at lambda - 372.1 nm and lambda =...
Questions in other subjects:


Computers and Technology, 15.01.2021 16:40


Mathematics, 15.01.2021 16:40

Physics, 15.01.2021 16:40

Mathematics, 15.01.2021 16:40




Mathematics, 15.01.2021 16:40

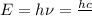 (Planck's equation)
(Planck's equation)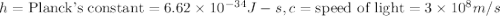
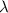 = wavelength of the photon with energy E in meters.
= wavelength of the photon with energy E in meters. = frequency of the photon with energy E in hertz.
= frequency of the photon with energy E in hertz.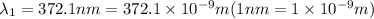
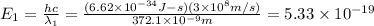 joules
joules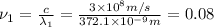 Hertz
Hertz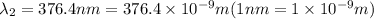
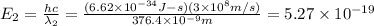 joules
joules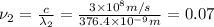 Hertz
Hertz

