
Chemistry, 17.11.2019 02:31 aaronw3743
Using the equation, 4fe + 3o2 imported asset 2fe2o3, if 8 moles of iron and oxygen from the air were available, how many moles of iron (iii) oxide would be produced?
4 moles
5 moles
6 moles
8 moles

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:40, Maryjasmine8001
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2


Chemistry, 22.06.2019 17:30, mwest200316
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
Using the equation, 4fe + 3o2 imported asset 2fe2o3, if 8 moles of iron and oxygen from the air were...
Questions in other subjects:

Health, 16.07.2019 01:30


Health, 16.07.2019 01:30



Social Studies, 16.07.2019 01:30



Social Studies, 16.07.2019 01:30

Health, 16.07.2019 01:30

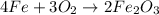
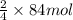 of iron oxide.
of iron oxide.

