
Chemistry, 18.09.2019 18:00 shelbycg02
What do colligative properties depend on? a) the identity of the solute b) the number of particles present in the solution c) the quantity of solvent in the solution d) none of these

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 12:30, kaliyab191
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2

Chemistry, 22.06.2019 13:00, cnfndbxbfbdb2031
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2
You know the right answer?
What do colligative properties depend on? a) the identity of the solute b) the number of particles...
Questions in other subjects:

Biology, 02.09.2019 15:30

Mathematics, 02.09.2019 15:30


History, 02.09.2019 15:30


English, 02.09.2019 15:30

History, 02.09.2019 15:30



English, 02.09.2019 15:30


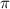 = osmotic pressure
= osmotic pressure


