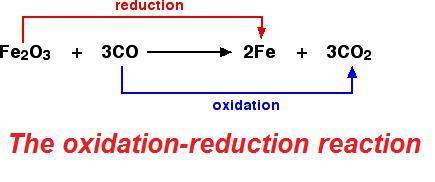
the reducing agent is reduced.


Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:00, graciewyatt6833
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 17:00, BREBRE8932
The biosphere of the earth is made up of what compound? organic or inorganic?
Answers: 3
You know the right answer?
What happens to a reducing agent during a redox reaction?
the reducing agent is reduced.
the reducing agent is reduced.
Questions in other subjects:

Mathematics, 22.07.2019 10:30





Mathematics, 22.07.2019 10:30



Mathematics, 22.07.2019 10:30

Mathematics, 22.07.2019 10:30