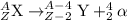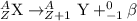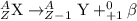Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:00, jaylanmahone223
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1


Chemistry, 22.06.2019 21:40, fatherbamboo
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
Alarge atom decays and emits a particle. after the reaction is complete, the atom’s mass has changed...
Questions in other subjects:


Health, 23.06.2021 06:40

Mathematics, 23.06.2021 06:40

English, 23.06.2021 06:40




Mathematics, 23.06.2021 06:40