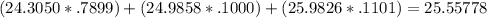Chemistry, 02.02.2020 22:56 RockieLuv7292
Magnesium has three naturally occurring isotopes: mg-24 with mass 24.3050 amu and a natural abundance of 78.99 %, mg-25 with mass 24.9858 amu and a natural abundance of 10.00 %, and mg-26 with mass 25.9826 amu and a natural abundance of 11.01 %.
calculate the atomic mass of magnesium.

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 16:50, TheOriginal2x
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
You know the right answer?
Magnesium has three naturally occurring isotopes: mg-24 with mass 24.3050 amu and a natural abundan...
Questions in other subjects:


History, 20.05.2020 05:02




Computers and Technology, 20.05.2020 05:02


Mathematics, 20.05.2020 05:02


English, 20.05.2020 05:02