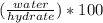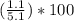Chemistry, 05.02.2020 13:01 andregijoe41
Asample of a hydrated compound has a mass of 5.1 g. during heating, it loses 1.1 g, leaving 4.0 g. what is the percentage by mass of water in the original hydrate?
6.11%
21.6%
27.5%
78.4%

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:10, cameronbeaugh
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 17:00, emma3216
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1


Chemistry, 22.06.2019 22:30, kiera2599
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
Asample of a hydrated compound has a mass of 5.1 g. during heating, it loses 1.1 g, leaving 4.0 g. w...
Questions in other subjects:

Mathematics, 27.01.2021 20:40

Chemistry, 27.01.2021 20:40







Physics, 27.01.2021 20:40