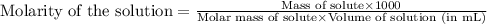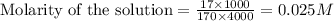Chemistry, 03.02.2020 00:49 hayleegahr
A4,000 ml solution of agno3 contains 17.00 g of solute in water. calculate the molarity (molar concentration) of the solution.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, NatalieKnows
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1

Chemistry, 22.06.2019 21:20, paatnguyyen
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2

Chemistry, 22.06.2019 21:20, carlydays4403
The organs inside the body and how they function together
Answers: 3

Chemistry, 22.06.2019 21:20, jordan2875
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1
You know the right answer?
A4,000 ml solution of agno3 contains 17.00 g of solute in water. calculate the molarity (molar conce...
Questions in other subjects:

Mathematics, 23.06.2019 07:00

History, 23.06.2019 07:00

History, 23.06.2019 07:00

History, 23.06.2019 07:00



Mathematics, 23.06.2019 07:00