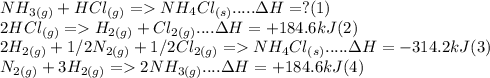- Calculate the Standard Enthalpy of the reaction below:
NH3(g) + HCI (g) → NH4Cl(s)
Using the...

Chemistry, 19.04.2021 01:00 maheshwarlall
- Calculate the Standard Enthalpy of the reaction below:
NH3(g) + HCI (g) → NH4Cl(s)
Using the following Enthalpy of Reactions:
2HCI(g) → H2(g) + Cl2(g)
AH = +184.6 KJ
2H2(g) + 1/2 N2(g) + 1/2 Cl2(g) → NH4Cl(s) deltaH = -314.4 kJ
N2(g) + 3 H2(g) → 2 NH3(g)
deltaH = +184.6 kJ

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, alexisgoss8091
Which traits do human embryos have that link them to a common ancestor with fish and reptiles? a. scales and tail b. gill slits and scales c. tail and gill slits d. hair and tail
Answers: 2
You know the right answer?
Questions in other subjects:



Mathematics, 15.01.2020 14:31

Physics, 15.01.2020 14:31



History, 15.01.2020 14:31