
Chemistry, 17.04.2021 19:40 maddy3lizabeth
Explain why each of these elements occur as diatomic molecules, Match the words in the left column to the appropriate blanks in the sentences on the right.
triple
double
octet
single
duet
When two hydrogen atoms pair together, they form a bond to achieve a complete which is a stable configuration.
When two halogens poir together, they form abond to achieve a complete which is a stable configuration
to achieve a completo
When two oxygen atoma pair together, they form abond to achieve a complete which is a stable configuration
When two nitrogen atomo poir together, they form abond to achieve a complete which is a stable configuration

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, madisonrosamond99
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 08:30, melikefood01
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1


Chemistry, 22.06.2019 17:00, emma3216
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1
You know the right answer?
Explain why each of these elements occur as diatomic molecules, Match the words in the left column t...
Questions in other subjects:

Mathematics, 03.08.2019 03:30

English, 03.08.2019 03:30


Social Studies, 03.08.2019 03:30

Mathematics, 03.08.2019 03:30

World Languages, 03.08.2019 03:30




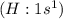 has one valence electron each, so sharing of electrons result in formation of single bond and the same electron configuration as the Helium and thus complete its duet.
has one valence electron each, so sharing of electrons result in formation of single bond and the same electron configuration as the Helium and thus complete its duet.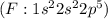 have 7 valence electron each, so sharing of electrons result in formation of single bond. The stable noble gas configuration is attained and thus complete its octet.
have 7 valence electron each, so sharing of electrons result in formation of single bond. The stable noble gas configuration is attained and thus complete its octet.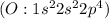 has six valence electrons each, so sharing of two electrons result in a double bond. The stable noble gas configuration is attained and thus complete its octet.
has six valence electrons each, so sharing of two electrons result in a double bond. The stable noble gas configuration is attained and thus complete its octet.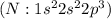 has 5 valence electrons, so sharing of three electrons result in a triple bond. The stable noble gas configuration is attained and thus complete its duet.
has 5 valence electrons, so sharing of three electrons result in a triple bond. The stable noble gas configuration is attained and thus complete its duet.

