
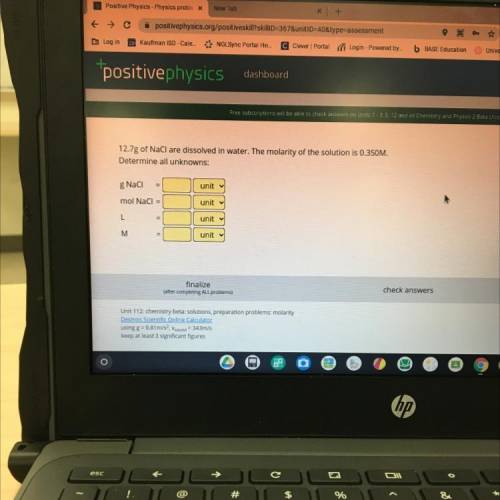
12.7g of NaCl are dissolved in water. The molarity of the solution is 0.350M.
Determine all unknowns:
g Naci
mol NaCl =
unity
L
unit
M
unity
finalize
(after completing ALL problems)
check answers
Unit 112: chemistry beta: solutions, preparation problems: molarity
Desmos Scientific Online Calculator

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 17:30, TheViperMlg23676
What causes most sediment to wash or fall into a river
Answers: 1

Chemistry, 23.06.2019 00:30, StayPuftMarshadowMan
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1
You know the right answer?
12.7g of NaCl are dissolved in water. The molarity of the solution is 0.350M.
Determine all unknown...
Questions in other subjects:


Physics, 30.03.2020 22:13






Advanced Placement (AP), 30.03.2020 22:13

Biology, 30.03.2020 22:13

Mathematics, 30.03.2020 22:13



