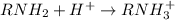Chemistry, 16.04.2021 03:20 makaylahunt
According to the Lewis model of acids and bases, amines behave as bases in water because . View Available Hint(s) According to the Lewis model of acids and bases, amines behave as bases in water because . the hydrogens will bond with hydroxide ions to make water they produce hydroxide ions they donate the hydrogens they have as hydrogen ions the unshared pair of electrons on the nitrogen can accept a proton

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, krystalhurst97
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

Chemistry, 22.06.2019 23:00, lufung8627
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

You know the right answer?
According to the Lewis model of acids and bases, amines behave as bases in water because . View Avai...
Questions in other subjects:

Mathematics, 20.09.2020 19:01

SAT, 20.09.2020 19:01







Mathematics, 20.09.2020 19:01

Mathematics, 20.09.2020 19:01

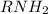 which has lone pair of electrons on nitrogen and thus is able to donate electrons to a lewis acid which is short of electrons. In other words nitrogen can accept a proton.
which has lone pair of electrons on nitrogen and thus is able to donate electrons to a lewis acid which is short of electrons. In other words nitrogen can accept a proton.