Why do weak acid-weak base reactions not go to completion?
there are not enough molecul...

Chemistry, 22.11.2019 04:31 jayycruz59
Why do weak acid-weak base reactions not go to completion?
there are not enough molecules in solution to react with each other.
the conjugate products are much weaker than the original acid and base.
neither a weak acid nor a weak base has a strong tendency to transfer h+ ions.
both weak acids and weak bases have a strong tendency to transfer h+ ions, so they cancel each other out.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:40, janelisse199820
Non renewable resources like petroleum eventually
Answers: 2


Chemistry, 22.06.2019 05:00, mahhvelousg97
You mix the pks of succinic acid are 4.21 and 5.64. how many gramsa graduate student at sdsu wants to measure the activity of a particular enzyme at ph 4.0. to buffer her reaction, she will use a buffer system based on one of the acids listed below, which acid is most appropriate for the experiment? of monosodium succinate (fw = 140 g/mol) and disodium succinate (fw = 162 g/mol) must be added to 1 l of water to produce a solution with a ph 5.28 and a total solute concentration of 100 mm? (assume the total volume remains 1 liter, answer in grams monosodium succinate, grams disodium succinate, respectively.) volumes of 0.05 m nah2po4 and 0.05 m na2hpo4 (pk's for phosphoric acid are 2.15, 6.82 and 12.38). which of the following best describes the resulting solution?
Answers: 2

Chemistry, 22.06.2019 05:50, zaleemawhite
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2
You know the right answer?
Questions in other subjects:


Mathematics, 06.04.2020 18:18


Mathematics, 06.04.2020 18:18

Chemistry, 06.04.2020 18:18

Social Studies, 06.04.2020 18:18

Computers and Technology, 06.04.2020 18:18



Mathematics, 06.04.2020 18:18

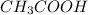 ) is a weak acid and it dissociates poorly in water.
) is a weak acid and it dissociates poorly in water.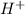 ions.
ions.

