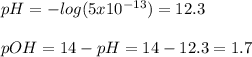Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:00, Isaiahtate053
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3


Chemistry, 22.06.2019 21:40, fatherbamboo
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 22.06.2019 22:10, zwbaby3693
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3
You know the right answer?
⦁Find the concentration of H+, OH-, PH and POH of 0.03 M of magnesium hydroxide which ionizes to the...
Questions in other subjects:

Mathematics, 10.04.2021 18:00

English, 10.04.2021 18:00


Mathematics, 10.04.2021 18:00


English, 10.04.2021 18:00

Mathematics, 10.04.2021 18:00


Social Studies, 10.04.2021 18:00

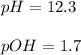
![[H^+]=5x10^{-13}M](/tpl/images/1260/4664/6a8b8.png)
![[OH^-]=0.02M](/tpl/images/1260/4664/8cf2b.png)
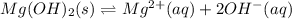
![\frac{1}{3} =\frac{x}{[Mg(OH)_2]}](/tpl/images/1260/4664/41f16.png)
![x=\frac{[Mg(OH)_2]}{3}=\frac{0.03M}{3}\\\\x= 0.01M](/tpl/images/1260/4664/c4205.png)
![[OH^-]=2x=2*0.01M=0.02M](/tpl/images/1260/4664/78e14.png)
![[H^+]=\frac{1x10^{-14}}{0.02}=5x10^{-13}M](/tpl/images/1260/4664/7fe92.png)