
Chemistry, 12.04.2021 20:40 Milanchik28
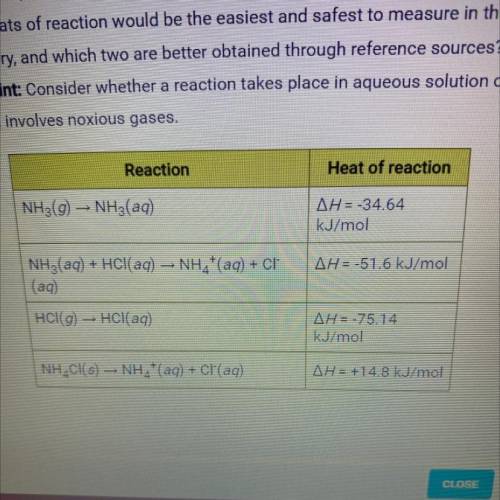
B. The heat of reaction for the process described in (a) can be determined by
applying Hess's law. The heats of reaction shown in the table below can be
obtained experimentally or looked up in tables of enthalpy data. Which two of
these heats of reaction would be the easiest and safest to measure in the
laboratory, and which two are better obtained through reference sources?
Why? Hint: Consider whether a reaction takes place in aqueous solution or
instead involves noxious gases.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:00, jamccoy3335
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1


Chemistry, 22.06.2019 11:00, jodonw5616
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1
You know the right answer?
B. The heat of reaction for the process described in (a) can be determined by
applying Hess's law....
Questions in other subjects:

History, 03.07.2019 18:00


Biology, 03.07.2019 18:00


Chemistry, 03.07.2019 18:00

History, 03.07.2019 18:00

Mathematics, 03.07.2019 18:00


Biology, 03.07.2019 18:00



